CAPABILITY
NEWS CENTER
APPLICATIONS
SOLUTIONS
SUPPORT
TOUCHPANEL BLOG
CONTACT US
Medical-Grade Touch Display OEM Guide: From Optical Bonding to ISO 13485, Building Highly Reliable TDM Solutions
11 Mar. 2026
This decision carries far greater consequences than it may appear. It directly impacts clinical workflow efficiency, diagnostic image accuracy, and — most critically — patient safety. Simply selecting a visually appealing screen based on a datasheet alone is no longer sufficient to meet the demands of real-world clinical applications.
This guide is designed to give OEM engineering and procurement teams a clear, actionable decision-making framework. We go beyond hardware specification comparisons to examine system integration, regulatory risk management, and Total Cost of Ownership (TCO) — helping you build a truly reliable medical-grade touchscreen display solution. Our objective is straightforward: to ensure your chosen solution delivers proven optical clarity, display consistency, and long-term reliability across critical care and point-of-care environments.
Integrating consumer-grade displays into medical devices is a shortcut filled with risk. The demands of clinical environments are fundamentally different from those of general commercial applications.
In radiology suites and operating rooms, clinicians interpret medical images in a fundamentally different way than viewing everyday photographs. They rely on extremely subtle grayscale gradations within CT scans, MRI images, or X-rays to identify clinically significant pathological indicators.
This is precisely why the DICOM GSDF (Grayscale Standard Display Function) was established. It defines a rigorous display standard with a single purpose: to ensure that any compliant display renders grayscale in a predictable, consistent manner — faithfully reproducing every detail captured in the image.
This also explains why consumer displays are simply not fit for purpose. They are engineered for vivid color reproduction and peak brightness — not for accurate grayscale fidelity. When grayscale rendering is inconsistent, subtle pathological features can be obscured, directly increasing the risk of misdiagnosis or missed findings. For medical devices, DICOM compliance is not an optional enhancement — it is a fundamental requirement for diagnostic accuracy and patient safety.
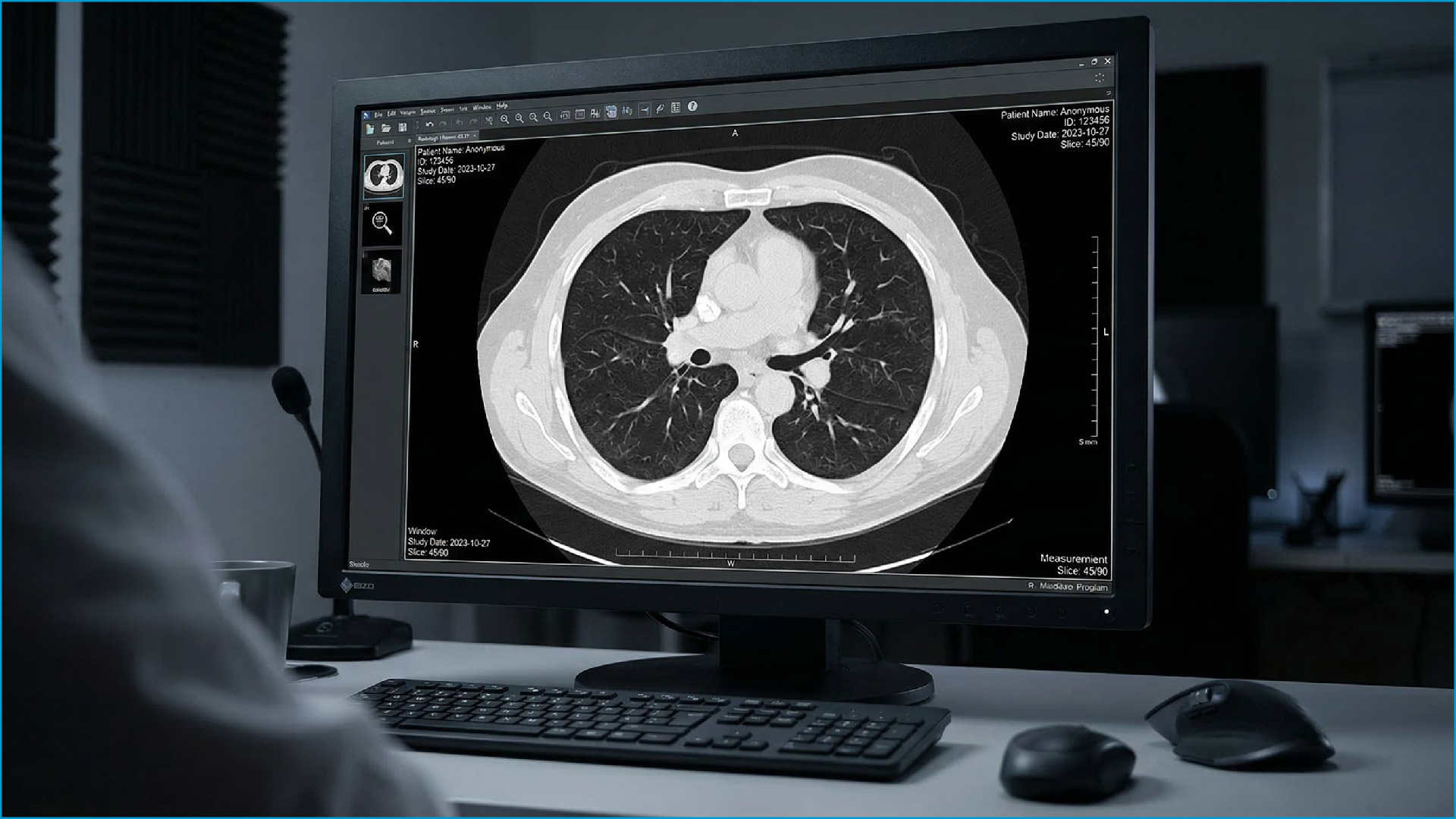
Medical devices are routinely expected to operate for years — often a decade or more — frequently running continuously for extended periods. Consumer displays may deliver impressive peak brightness at the outset, but their luminance degradation rate far exceeds what medical applications can tolerate. As brightness declines, images become dim and contrast is reduced, forcing clinicians to interpret under increasingly compromised conditions.
This instability ultimately translates into real operational costs. Frequent display recalibration, premature replacement, and the potential diagnostic risks associated with degraded image quality all far outweigh any initial hardware savings. A qualified medical-grade display must incorporate luminance retention over thousands of hours of use as a core design requirement — not an afterthought.
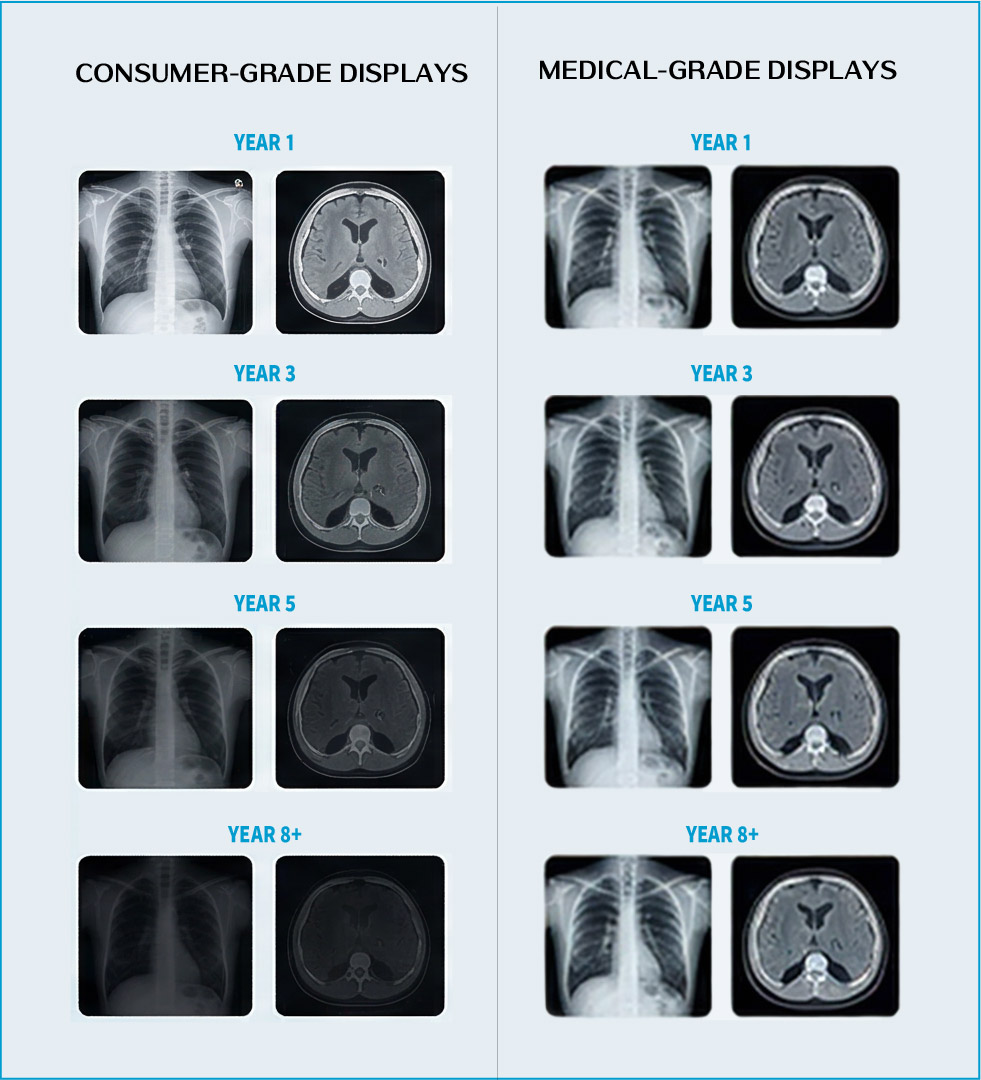
When reviewing a CT image, a clinician requires absolute focus. If the display exhibits inconsistent brightness between the center and corners of the panel, it introduces a form of visual noise — subtly disrupting concentration and reducing diagnostic efficiency. This luminance non-uniformity may force clinicians to repeatedly pan or zoom to verify details, and can even obscure pathological findings located in dimmer regions of the display.
Commercial displays typically apply relatively loose luminance uniformity calibration tolerances. Medical-grade displays, by contrast, must control brightness variation across the entire panel within a tightly defined range — ensuring every region of the image is rendered clearly and consistently.
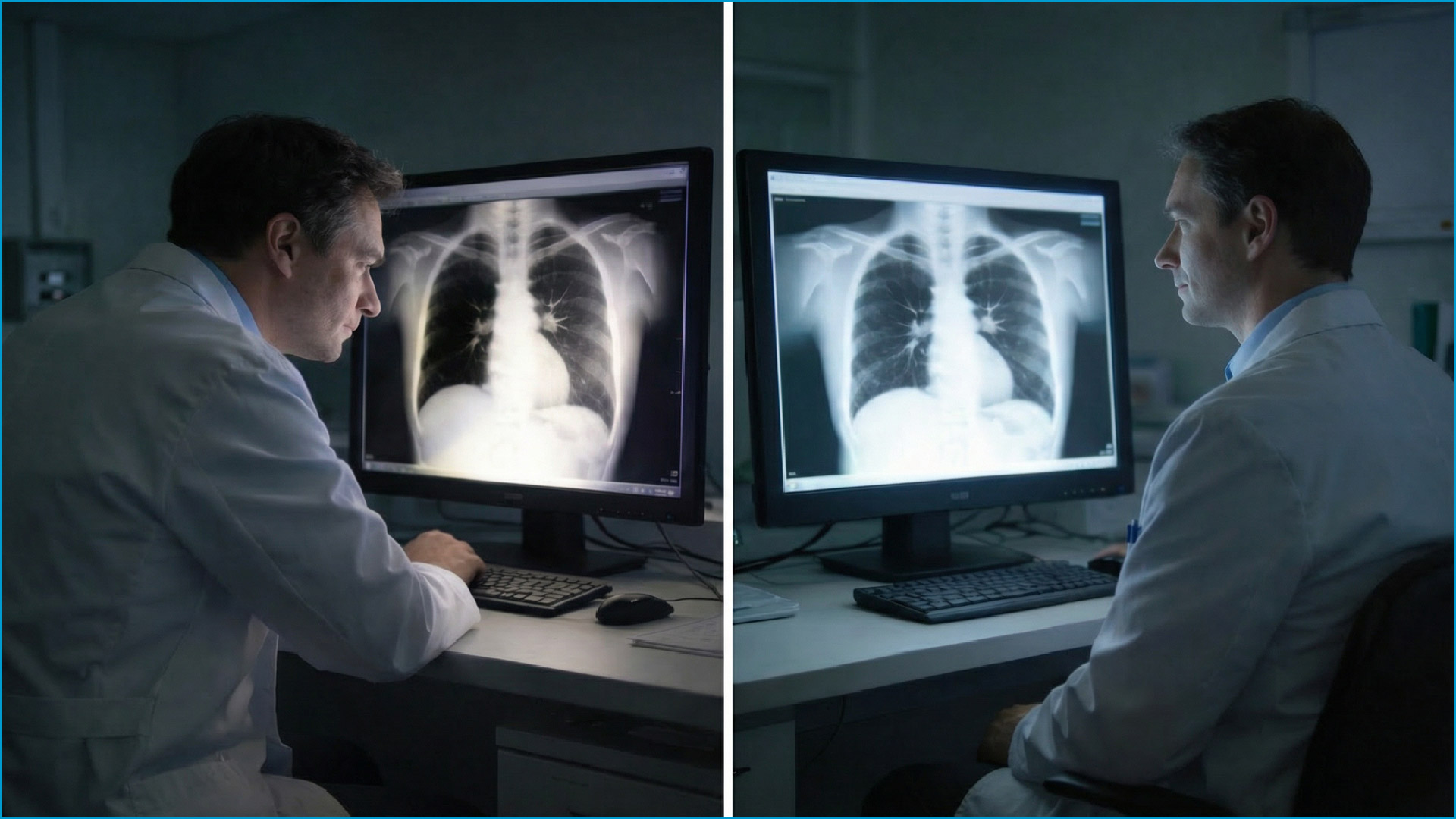
In operating rooms and multidisciplinary team (MDT) consultations, clinical teams need to view the same display from varying angles, or compare images across multiple screens simultaneously. If a display exhibits significant color shift or contrast degradation when viewed off-axis, it directly undermines the accuracy of team communication. Similarly, color or luminance discrepancies between two side-by-side displays can compromise diagnostic consistency.
These challenges demand that medical-grade displays employ higher-specification panel technology and more rigorous calibration processes — ensuring stable, reliable image performance in wide-angle viewing and multi-display configurations alike.
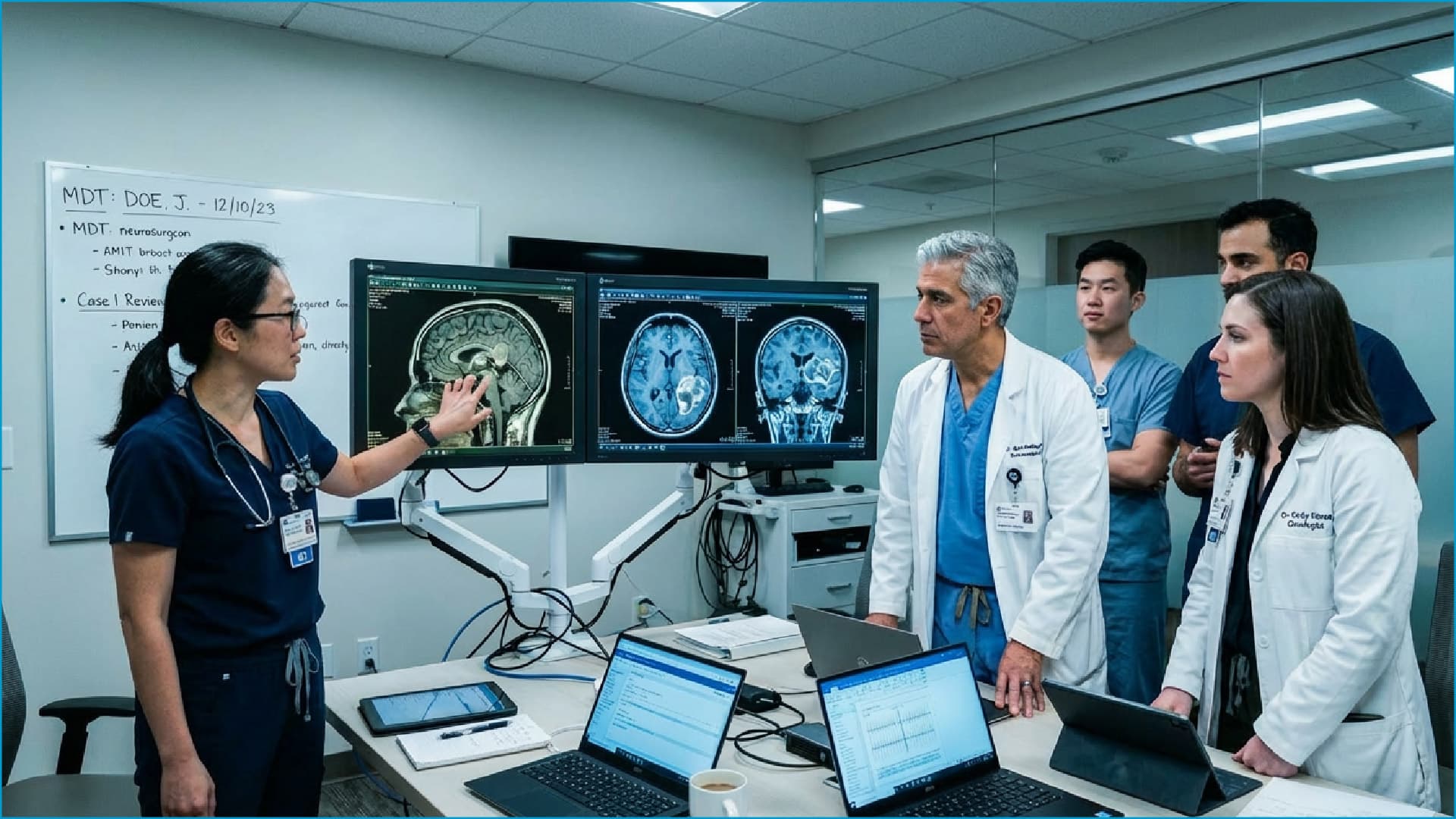
The user interface at the front end of a medical device is the primary point of interaction between the clinician and the system. Its design must address the multiple, simultaneous demands of clinical environments.
Continuous Operation in High-Pressure Environments
Whether in emergency departments, intensive care units (ICUs), or operating rooms, devices may be required to operate reliably 24/7 without interruption.
Repeated Chemical Cleaning and Disinfection
The display surface must withstand prolonged exposure to a wide range of medical-grade disinfectants and cleaning agents without functional degradation or surface deterioration.
Accurate Touch Response Through Medical Gloves
Clinical staff operate almost exclusively while wearing latex or nitrile gloves. The touchscreen system must maintain high sensitivity and positional accuracy under these conditions.
Visibility Under Complex Ambient Lighting
Surgical shadowless lights and varied ambient light sources can cause significant glare and reflections on display surfaces, directly interfering with image interpretation.
Sourcing a high-quality display panel alone cannot address all of the challenges above.
This is precisely why integrated TDM (Touch Display Module) solutions
have become the preferred choice for modern medical device OEMs.
The core concept of a TDM (Touch Display Module) is to pre-integrate the display panel, touch sensor, cover glass, and optical bonding into a single, fully validated modular solution. This approach delivers three direct benefits:
Traditionally, OEMs integrating separate display panels and touch sensors from different vendors frequently encounter issues such as tolerance mismatches, touch sensitivity failures, signal interference, or degraded display quality. A TDM resolves these variables within the module itself — significantly reducing system-level integration risk.
Adopting a pre-validated TDM module allows OEM engineering teams to eliminate extensive testing and tuning cycles, redirecting engineering effort toward core system development and accelerating time-to-market.
A modular design and manufacturing process helps ensure that every production batch meets the same high standards of quality and reliability — reducing production variability and improving overall yield.
For OEMs, adopting a TDM is not merely a technology upgrade — it is an effective strategy for risk management and operational efficiency.
Within a TDM module, optical bonding technology plays a critical role. It directly determines the display's performance across three essential dimensions: visual clarity, touch precision, and long-term durability.
Enhanced Visibility: Eliminating Glare and Reflections
Conventional air-gap bonding leaves an air layer between the display panel and the cover glass. Because air has a different refractive index from both glass and the panel, it generates significant internal reflections under bright light — producing glare and substantially reducing contrast and image clarity.
Optical bonding eliminates this air layer entirely by filling it with an optically clear adhesive (OCA). Since the adhesive's refractive index closely matches that of glass, light passes through with minimal reflection. For clinicians who need to interpret images clearly under surgical lighting, this technology ensures that image detail is not compromised by ambient light — providing a direct improvement in diagnostic accuracy.
Parallax Elimination: Achieving "Touch-Where-You-See" Precision
A further drawback of air-gap bonding is optical parallax. Because the air layer separates the touch surface from the display pixels, the user's line of sight, the touch contact point, and the on-screen pixel are not perfectly aligned — particularly noticeable when viewing from an angle, where the touch response appears offset from the intended target.
Optical bonding eliminates this air gap, fusing the touch layer and display layer into direct contact and completely resolving parallax. For medical applications requiring fine selection or drag operations — such as parameter adjustment on ultrasound equipment or landmark annotation in surgical navigation systems — this significantly enhances operational intuitiveness and clinician confidence.
Risk Mitigation: Preventing Condensation and Fogging
Medical devices are frequently moved between environments with different temperature and humidity conditions — for example, from a temperature-controlled storage room into a warm, humid operating theater. In an air-gap bonded structure, moisture trapped within the air layer can condense into droplets or fog when exposed to temperature differentials, impairing visibility and, over time, potentially damaging internal electronic components.
Optical bonding replaces the air layer with a solid adhesive material, fundamentally eliminating the risk of condensation and fogging — ensuring the TDM module maintains stable optical performance and reliability across all operating environments.
Structural Reinforcement: Withstanding the Demands of Mobile and Impact-Prone Use
Mobile medical carts and portable patient monitors inevitably encounter vibration and impact during use. Optical bonding bonds the cover glass, touch layer, and display panel into a single, rigid assembly — significantly increasing the structural integrity and impact resistance of the entire module, and effectively extending service life in demanding clinical environments.
| Evaluation Criteria | Optical Bonding | Air-Gap Bonding |
|---|---|---|
| Visibility | High contrast, low reflectance — suitable for complex ambient lighting conditions | Significant glare; suitable only for stable, controlled indoor lighting |
| Touch Accuracy | Zero parallax; touch response is intuitive and precise | Parallax present; impacts fine-touch operation experience |
| Environmental Tolerance | No condensation or fogging risk; suitable for environments with temperature and humidity variation | Condensation and fogging risk present; may damage internal components over time |
| Structural Integrity | Robust construction; high resistance to impact and vibration | Relatively fragile structure; lower impact resistance |
| Cost Consideration | Higher initial manufacturing cost, but superior long-term TCO | Lower initial procurement cost, but higher downstream maintenance and failure risk |
For medical devices where long-term reliability and optimal user experience are paramount, optical bonding commands a higher initial investment — but the stability and durability it delivers will significantly reduce Total Cost of Ownership over the full product lifecycle.
Once the decision to adopt a TDM with optical bonding has been made, OEM teams must still make the right choices in technical process, structural design, and supplier selection to ensure manufacturability and final product quality.
Applied as a pre-formed adhesive film, OCA offers uniform thickness, high process stability, minimal bubble formation, and relatively straightforward rework. Its limitations include higher material cost and limited ability to compensate for larger tolerance gaps between the panel and cover glass — making it less suitable for large-format or irregular surface bonding applications.
Applied as a liquid and cured via UV light or heat, LOCA can perfectly fill irregular surfaces and larger tolerance gaps, making it better suited for large-format displays. However, its process window is narrow — demanding extremely tight control over environmental cleanliness, adhesive dispensing uniformity, and curing parameters. It is prone to bubble formation and adhesive overflow, and rework after curing is exceptionally difficult.
OEM Selection Guidance
→ If your device demands high visual consistency and is 15 inches or smaller, OCA is the more robust, lower-risk choice.
→ If your device features a large-format display or requires the adhesive to compensate for larger structural tolerances, LOCA offers greater flexibility — provided your supplier has demonstrated, mature process control capabilities.
Surface treatment selection for medical touchscreen displays must strike the right balance between optical performance and long-term durability.
Achieved through etching or coating to create a micro-textured surface that scatters ambient light and reduces glare. Well-suited for high-ambient-light environments such as operating rooms. Note that low-quality AG treatments can reduce image sharpness, and the coating's abrasion resistance must be sufficient to withstand repeated chemical wiping over time.
Multi-layer optical thin films deposited on the glass surface use optical interference principles to suppress reflections. AR coatings maximally preserve image contrast and clarity, making them ideal for high-precision diagnostic imaging applications. Chemical resistance of the AR coating is a critical evaluation criterion.
A hydrophobic and oleophobic nano-coating that prevents fingerprints and contaminants from adhering to the surface, while facilitating easy cleaning. This is essential for the day-to-day maintenance of touchscreen displays in clinical settings.
A functional coating applied to material surfaces — including glass, plastic, and metal — that inhibits or eliminates bacteria, mold, and other microorganisms, reducing the risk of contamination and cross-infection in clinical environments.
OEM Selection Guidance
Define Your Requirements Clearly
Based on your device's specific use environment, require your supplier to provide a surface treatment solution that combines multiple functional properties — and to furnish complete abrasion resistance and chemical compatibility test reports.
The performance of a TDM module also depends on how well it integrates with the overall system architecture.
Electromagnetic Interference (EMI) Shielding
Medical environments are dense with electronic equipment, and the electromagnetic fields they generate can interfere with projected capacitive (PCAP) touch signals — causing touch drift or complete touch failure. TDM modules must incorporate robust shielding design and proper grounding architecture.
System Synchronization
The touch controller's coordinate reporting rate must be well-matched to the system's display refresh rate to deliver a smooth, low-latency user experience.
Comprehensive System Integration Testing
Prior to mass production, the TDM module must be installed in the final product prototype and subjected to full stress testing — simulating all anticipated clinical use scenarios to validate long-term stability and reliability.
For medical devices, meeting specifications on paper is not enough. All performance claims must be validated and supported by auditable documentation.
The most distinctive challenge for medical touchscreen displays is the requirement for frequent chemical cleaning. A complete solution must address this through three layers of design:
Material Compatibility
Every material in the stack — from cover glass and optical adhesive to surface coatings — must be selected for resistance to the most common medical-grade disinfectants, including isopropyl alcohol (IPA) and quaternary ammonium compounds (QACs).
Structural Sealing
Optimized edge sealing design prevents cleaning fluids from ingressing into the module interior and causing long-term damage.
Verifiable Test Protocols
Clearly defined test methodologies must be established — specifying which disinfectants, applied at what pressure, over how many wipe cycles — after which no degradation in appearance, optical performance, or touch functionality is permitted. This transforms the vague claim of "durability" into quantifiable, auditable quality evidence.
ISO 13485 is the quality management system standard specifically designed for the medical device industry. Selecting a TDM supplier that holds ISO 13485 certification — with a scope covering both design and manufacturing — delivers the following direct benefits to OEMs:
Reduced Regulatory Risk
The supplier's design, development, change management, and manufacturing processes already conform to the traceability and documentation requirements mandated by medical device regulations — significantly streamlining your FDA 510(k) or CE Technical File submissions.
Supply Chain Stability
ISO 13485-compliant suppliers maintain more robust quality control and risk management processes, effectively reducing the risk of delivery delays or product recalls caused by quality failures.
Improved Project Efficiency
You avoid the overhead of auditing a supplier unfamiliar with medical industry requirements. Both parties can collaborate efficiently within a shared quality framework and common regulatory language.
For us, this is more than a certificate — it is a commitment to our customers. It demonstrates that we understand the exacting standards of the medical device industry, and that we have embedded this quality management system into every stage of our operations, from design and development through to manufacturing.
This proactive investment enables us to move beyond the role of a component supplier — becoming a strategic partner capable of working alongside you to navigate the challenges of bringing medical devices to market.
A display solution that appears inexpensive at the point of purchase will often extract far greater hidden costs over the product lifecycle through the following:
Higher Maintenance and Replacement Costs
Frequent servicing and component replacement driven by luminance degradation, touch failure, or structural damage.
Higher Operational Downtime Costs
Device downtime caused by display failures, resulting in lost clinical efficiency and customer complaints.
Higher Quality Management Costs
Additional engineering and quality assurance resources required to manage an inconsistent or unreliable supplier.
Selecting a high-quality, high-reliability TDM solution carries a higher initial procurement cost — but it delivers longer product service life, lower maintenance requirements, and a more consistent end-user experience. Evaluated through the lens of TCO, this is the genuinely cost-effective decision.
Selecting the right display and touch interface for a medical device is a high-stakes strategic decision. It directly defines product performance and user experience — and ultimately shapes your brand reputation and competitive position in the market.
By adopting an integrated TDM approach and incorporating optical bonding, reliability validation, and ISO 13485-certified supplier qualification as core evaluation criteria, your team can effectively reduce project risk, accelerate development timelines, and build products capable of delivering sustained, reliable performance in the most demanding clinical environments.
This is how you deliver on your quality promise to customers — and how you make an investment that protects the long-term value of your business.
DICOM Part 14 (Grayscale Standard Display Function) compliance is the foundation of any diagnostic imaging display. It ensures consistent grayscale rendering, preventing subtle pathological features from being obscured by inconsistent luminance response. This is a fundamental requirement for diagnostic accuracy and patient safety — not an optional enhancement.
Optical bonding eliminates the air gap between the display panel and cover glass — reducing reflectance, improving contrast and image clarity, and eliminating parallax for precise touch response. It also prevents condensation and fogging, and significantly enhances structural integrity against vibration and impact.
A TDM pre-integrates the display panel, touch sensor, cover glass, and optical bonding into a single validated module — reducing system integration risk, accelerating development timelines, and ensuring consistent mass production quality. It is an effective strategy for both risk management and operational efficiency.
OCA (solid film adhesive) offers high process stability and is best suited for smaller displays; LOCA (liquid adhesive) accommodates larger formats and structural tolerances but demands tighter process control. Selection should be based on display size and the supplier's demonstrated manufacturing capabilities.
ISO 13485 certification confirms that a supplier has established medical-grade design and manufacturing capabilities — reducing regulatory risk, ensuring supply chain stability, and improving project efficiency. Selecting a certified supplier simplifies FDA 510(k) submissions and CE Technical File preparation.
While a high-quality TDM carries a higher initial procurement cost, it reduces downstream maintenance and replacement expenses, minimizes operational downtime risk, and delivers a more consistent user experience over a longer product service life — making it the more cost-effective choice when evaluated over the full product lifecycle.
We Help You Reduce Integration Risks and Accelerate Time-to-Market